Role of active transport of potassium to leaves in the mechanisms of tolerance to salinity in common bean (Phaseolus vulgaris L.)
DOI:
https://doi.org/10.15835/nsb12210751Keywords:
bean cultivars; potassium transport; proline; salt stress; sodium exclusionAbstract
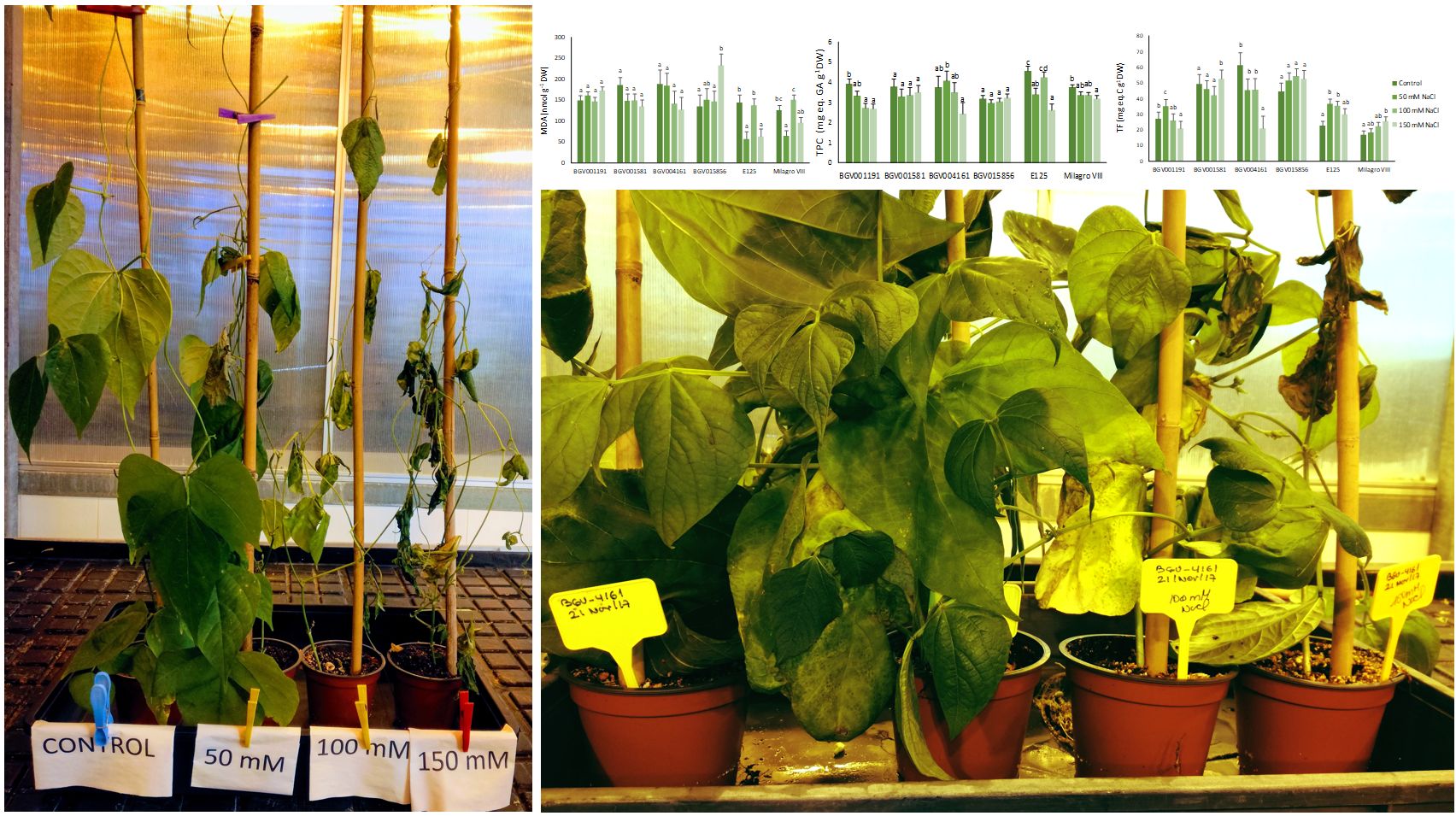
Biochemical markers are of great utility in screening for salt tolerance of crops. In common beans (Phaseolus vulgaris), lower levels of proline under stress have been associated with a better stress resistance of cultivars. In the present study, the responses to salinity have been analysed in six cultivars of common beans: four local landraces from Spain and two experimental lines from Cuba. Proline was used for ranking the relative tolerance of the cultivars, confirming a previous study which reported as more stress-tolerant two of the Spanish landraces. Total soluble sugars concentrations varied with treatments and between genotypes, but it was difficult to assess their role in stress tolerance of the analysed plants. Sodium concentration in leaves was the lowest in one of the two salt-resistant cultivars, and potassium did not vary or even increased under salt stress in all of them, except for the most susceptible one, where a drop of this cation was registered under 150 mM NaCl. Changes in malondialdehyde (MDA) contents did not indicate salt-induced membrane peroxidation resulting from secondary oxidative stress; consequently, accumulation of total phenolic compounds and flavonoids, as an antioxidant defence mechanism, was not detected. These results highlight the reliability of using proline as a biochemical marker of salt stress in common beans and the importance of the mechanism related to potassium transport to leaves in conferring stress tolerance to some common bean cultivars.
Metrics
References
Abdallah MMS, Abdelgawad ZA, El-Bassiouny HMS (2016). Alleviation of the adverse effects of salinity stress using trehalose in two rice varieties. South African Journal of Botany 103:275-282. https://doi.org/10.1016/j.sajb.2015.09.019
Al Hassan M, Morosan M, López-Gresa MP, Prohens J, Vicente O, Boscaiu M (2016). Salinity-induced variation in biochemical markers provides insight into the mechanisms of salt tolerance in common (Phaseolus vulgaris) and runner (P. coccineus) beans. International Journal of Molecular Sciences 17:1582. https://doi.org/10.3390/ijms17091582
Apel K, Hirt H (2004). Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annual Review of Plant Biology 55:373-399. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Arteaga S, Yabor L, Díez, MJ, Prohens J, Boscaiu M, Vicente O (2020). The use of proline in screening for tolerance to drought and salinity in common bean (Phaseolus vulgaris L.) genotypes. Agronomy 10:817. https://doi.org/10.3390/agronomy10060817
Arteaga S, Yabor L, Torres J, Solbes E, Muñoz E, Díez MJ … Boscaiu M. (2019). Morphological and agronomic characterisation of Spanish landraces of Phaseolus vulgaris L. Agriculture 9:149. https://doi.org/10.3390/agriculture9070149
Ashraf M, Iram A (2005). Drought stress induced changes in some organic substances in nodules and other plant parts of two potential legumes differing in salt tolerance. Flora 200:535-546. https://doi.org/10.1016/j.flora.2005.06.005
Bahena-Betancourt L, Macias-Rodríguez L, Bayuelo-Jiménez JS (2006). Contents of carbohydrates in salt-stressed Phaseolus species; Annual report of the bean improvement cooperative. United States Department of Agriculture: Fort Collins, CO, USA pp 233-234.
Bates LS, Waldren RP, Teare ID (1973). Rapid determination of free proline for water stress studies. Plant and Soil 39:205-207.
Bayuelo-Jiménez JS, Jasso-Plata N, Ochoa I (2012). Growth and physiological responses of Phaseolus species to salinity stress. International Journal of Agronomy 80:207-222. https://doi.org/10.1155/2012/527673
Blainski A, Lopes GC, Palazzo de Mello JC (2013). Application and analysis of the Folin Ciocalteu method for the determination of the total phenolic content from Limonium brasiliense L. Molecules 18:6852-6865. https://doi.org/10.3390/molecules18066852
Broughton WJ, Hernández G, Blair M, Beebe S, Gepts P, Vanderleyden J (2003). Beans (Phaseolus spp.)-Model food legumes. Plant and Soil 252:55-128. https://doi.org/ 10.1023/A:1024146710611
Cárdenas-Avila M, Verde-Star J, Maiti R, Foroughbakhch R, Gámez-González H, Martínez-Lozano S, ... Morales-Vallarta M (2006). Variability in accumulation of free proline on in vitro calli of four bean (Phaseolus vulgaris L.) cultivars exposed to salinity and induced moisture stress. Phyton 75:103-108.
Daliakopoulos IN, Tsanis IK, Koutroulis AG, Kourgialas N, Varouchakis EA, Karatzas GP, Ritsema CJ (2016). The threat of soil salinity: A European scale review. Science of Total Environment 573:727–739. https://doi.org/10.1016/j.scitotenv.2016.08.177
Del Río LA, Palma JM, Sandalio LM, Corpas FJ, Pastori GM, Bueno P, López‐Huertas E (1996). Peroxisomes as a source of superoxide and hydrogen peroxide in stressed plants. Biochemical Society Transactions 24:434-438. https://doi.org/10.1042/bst0240434
Domínguez A, Yunel Pérez Y, Alemán S, Sosa M, Fuentes, L.; Darias, R … Sosa D (2014). Respuesta de cultivares de Phaseolus vulgaris L. al estrés por sequía. Biotecnologia Vegetal 14:29-36.
Dubois M, Gilles KA, Hamilton JK, Reberd PA, Smith F (1956). Colorimetric method for determination of sugars and related substances. Analytical Chemistry 28:350-356. https://doi.org/10.1021/ac60111a017
Dwivedi SL, Ceccarelli S, Blair MW, Upadhyaya HD, Are AK, Ortiz R (2016). Landrace germplasm for improving yield and abiotic stress adaptation. Trends in Plant Science 21:31-42. https://doi.org/10.1016/j.tplants.2015.10.012
Fita A, Rodríguez-Burruezo A, Boscaiu M, Prohens J, Vicente O (2015). Breeding and domesticating crops adapted to drought and salinity: A wew paradigm for increasing food production. Frontiers in Plant Science 6:978. https://doi.org/10.3389/fpls.2015.00978
Flowers TJ, Colmer TD (2008). Salinity tolerance in halophytes. New Phytologist 179:945-963. doi: https://doi.org/10.1111/j.1469-8137.2008.02531.x
Flowers TJ, Hajibagheri MA, Clipson NJW (1986). Halophytes. The Quarterly Review of Biology 61:313-335.
Gama P, Inanaga S, Tanaka K, Nakazawa R (2007). Physiological response of common bean (Phaseolus vulgaris L.) seedlings to salinity stress. African Journal of Biotechnology 6:79-88.
Gepts P, Debouck DG (1991). Origin, domestication, and evolution of the common bean, Phaseolus vulgaris. In: Van Schoonhoven A, Voysest O (Eds) Common beans: research for crop improvement. Cab International Wallingford UK pp 7-53.
Ghanbari AA, Mousavi SH, Mousapou Gorji A, Rao I (2013). Effects of water stress on leaves and seeds of bean (Phaseolus vulgaris L.). Turkish Journal of Field Crops 18:73-77. https://doi.org/10.1016/0378-4290(89)90054-3
Gierth M, Mäser P (2007). Potassium transporters in plants- Involvement in K+ acquisition, redistribution and homeostasis. FEBS Letters 581:2348–2356. https://www.altmetric.com/details/1739783
Gil R, Boscaiu MT, Lull C, Bautista I, Lidón AL, Vicente O (2013). Are soluble carbohydrates ecologically relevant for salt tolerance in halophytes? Functional Plant Biology 40:805-818. https://doi.org/10.1071/FP12359
Grigore MN, Boscaiu M, Vicente O (2011). Assessment of the relevance of osmolyte biosynthesis for salt tolerance of halophytes under natural conditions. European Journal of Plant Science and Biotechnology 5:12-19.
Hayat S, Hayat Q, Alyemeni MN, Wani AS, Pichtel J, Ahmad A (2012). Role of proline under changing environments: a review. Plant Signaling and Behaviour 7:1456-1466. https://doi.org/10.4161/psb.21949
Herrera Flores TS, Ortíz Cereceres J, Delgado Alvarado A, Acosta Galleros JA (2012). Growth and, proline and carbohydrate content of bean seedlings subjected to drought stress. Revista Mexicana de Ciencias Agricolas 3:713-725.
Hoagland DR, Arnon DI (1950). The water culture method for growing plants without soil. California Agricultural Experimental Station Circular No. 347, University of California, Berkeley pp 1-32.
Hodges DM, De Long JM, Forney CF, Prange RK (1999). Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 207:604-611. https://doi.org/10.1007/s004250050524
Hurtado M, Vilanova S, Plazas M, Gramazio P, Andújar I, Herraiz FJ, Prohens J (2014). Enhancing conservation and use of local vegetable landraces: the Almagro eggplant (Solanum melongena L.) case study. Genetic Resources and Crop Evolution 61:787-795. https://doi.org/10.1007/s10722-013-0073-2
Hussain TM, Chandrasekhar T, Hazara M, Sultan Z, Saleh BK, Gopal GR (2008). Recent advances in salt stress biology – a review. Biotechnology and Molecular Biology Reviews 3(1):008-013.
Jiménez-Bremont JF, Becerra-Flora A, Hernández-Lucero E, Rodríguez-Kessler M. Acosta-Gallegos J, Ramírez Pimentel J (2006). Proline accumulation in two bean cultivars under salt stress and the effect of polyamines and ornithine. Biologia Plantarum 50:763-766. https://doi.org/10.1007/s10535-006-0126-x
Kavi Kishor P, Sreenivasulu N (2014). Is proline accumulation per se correlated with stress tolerance or is proline homeostasis a more critical issue? Plant Cell and Environment 37:300-311. https://doi.org/10.1111/pce.12157
Kaymakanova M, Stoeva N (2008). Physiological reaction of bean plants (Phaseolus vulgaris L.) to salt stress. General and Applied Plant Physiology 34:177-188.
Kozminska A, Al Hassan M, Hanus-Fajerska E, Naranjo MA, Boscaiu M, Vicente O (2018). Comparative analysis of water deficit and salt tolerance mechanisms in Silene. South African Journal of Botany 117:193-206. https://doi.org/10.1016/j.sajb.2018.05.022
Larcher W (2003). Physiological plant ecology. Ecophysiology and stress physiology of functional groups. Springer (4th ed), Berlin.
Mackay CE, Hall C, Hofstra G, Fletcher RA (1990). Uniconazole-induced changes in abscisic acid, total amino acids, and proline in Phaseolus vulgaris. Pesticide Biochemistry and Physiology 37:74-82. https://doi.org/10.1016/0048-3575(90)90110-N
Mercer KL, Perales HR (2010). Evolutionary response of landraces to climate change in centers of crop diversity. Evolutionary Applications 3(5-6):480‐493. https://doi.org/10.1111/j.1752-4571.2010.00137.x
Misra N, Gupta AK (2005). Effect of salt stress on proline metabolism in two high yielding genotypes of green gram. Plant Science 169:331-339. https://doi.org/10.1016/j.plantsci.2005.02.013
Morosan M, Al Hassan M, Naranjo M, Lopez-Gresa MP, Vicente O (2017). Comparative analysis of drought responses in Phaseolus vulgaris (common bean) and P. coccineus (runner bean) cultivar. EuroBiotech Journal 1:247-252. https://doi.org/10.24190/ISSN2564-615X/2017/03.09
Nagesh B, Devaraj V (2008). High temperature and salt stress response in French bean (Phaseolus vulgaris). Australian Journal of Crop Science 2:40-42.
Pinheiro C, Baeta JP, Pereira AM, Dominguez H, Ricardo C (2007). Mineral elements correlations in a Portuguese germplasm collection of Phaseolus vulgaris. Integrating legume biology for sustainable agriculture. In: Proceedings of the 6th European Conference on Grain Legumes, Lisbon 2007, pp 125-126.
Rahma LZ, Fawzia TB, Zineb T (2019). Phytochemical potential and antioxidant activity of Phaseolus vulgaris L. (Var, Sidi Ferredj) in the face of salt stress. South Asian Journal of Experimental Biology 9:2. http://sajeb.org/index.php/sajeb/article/view/20675
Rana V, Ram S, Nehra K (2017). Proline biosynthesis and its role in abiotic stress. International Journal of Agricultural Research, Innovation and Technology 6(3):473-478.
Raza A, Razzaq A, Zou X, Mehmood S, Zou X, Lv Y, Xu J (2019). Impact of climate change on crops adaptation and strategies to tackle its outcome: A review. Plants 8(2):34. https://doi.org/10.3390/plants8020034
Rendón-Anaya M, Montero-Vargas JM, Saburido-Álvarez S, Vlasova A, Capella-Gutierrez S, Ordaz-Ortiz JJ … Herrera-Estrella A (2017). Genomic history of the origin and domestication of common bean unveils its closest sister species. Genome Biology 18:60. https://doi.org/10.1186/s13059-017-1190-6
Rosa M, Prado C, Podazza G, Interdonato R, González JA, Hilal M, Prado FE (2009). Soluble sugars-Metabolism, sensing and abiotic stress. A complex network in the life of plants. Plant Signaling & Behavior 4(5):388-393. https://doi.org/10.4161/psb.4.5.8294.
Rosales MA, Ocampo O, Rodríguez-Valentín R, Olvera-Carrillo Y, Acosta-Gallegos J, Covarrubias AA (2012). Physiological analysis of common bean (Phaseolus vulgaris L.) cultivars uncovers characteristics related to terminal drought resistance. Plant Physiology and Biochemistry 56:24-34. https://doi.org/10.1016/j.plaphy.2012.04.007
Schiop ST, Al Hassan M, Sestras AF, Boscaiu M, Sestras RE, Vicente O (2015). Identification of salt stress biomarkers in Romanian Carpathian populations of Picea abies (L.) Karst. PLoS ONE 10(8):e0135419. https://doi.org/10.1371/journal.pone.0135419
Seemann JR, Critchley C (1985). Effects of salt stress on the growth, ion content, stomatal behaviour and photosynthetic capacity of a salt-sensitive species, Phaseolus vulgaris L. Planta 164:151-162. https://doi.org/10.1007/BF00396077
Shabala S, Cuin TA (2007). Potassium transport and plant salt tolerance. Physiologia Plantarum 133:651-669.
Szabados L, Savouré A (2010). Proline: A multifunctional amino acid. Trends in Plant Science 15:89-97. https://doi.org/10.1016/j.tplants.2009.11.009.
Taïbi K, Taïbi F, Abderrahim L, Ennajah A, Belkhodja M, Mulet JM (2016). Effect of salt stress on growth, chlorophyll content, lipid peroxidation and antioxidant defence systems in Phaseolus vulgaris L. South African Journal of Botany 105:306-312. https://doi.org/10.1016/j.sajb.2016.03.011
Tazuke A, Wada T, Kanazawa T (2009). Effects of NaCl salinity on the sugar metabolism of common bean (Phaseolus vulgaris L.) cv. ‘Tsurunashi Marusaya Kurosando’ fruit grown in solution culture. Journal of the International Society for Southeast Asian Agricultural Sciences 15:32-43.
Tester M, Davenport R (2003). Na+ tolerance and Na+ transport in higher plants. Annals of Botany 91:503-527. https://doi.org/10.1093/aob/mcg058
Türkan I, Demiral T (2009). Recent developments in understanding salinity tolerance. Environmental and Experimental Botany 67:2-9. https://doi.org/10.1016/j.envexpbot.2009.05.008
Volkov V, Wang B, Dominy PJ, Fricke W, Amtmann A (2003). Thellungiella halophila, a salt-tolerant relative of Arabidopsis thaliana, possesses effective mechanisms to discriminate between potassium and sodium. Plant Cell and Environment 27:1-14. https://doi.org/10.1046/j.0016-8025.2003.01116.x
Weimberg R (1987). Solute adjustments in leaves of two species of wheat at two different stages of growth in response to salinity. Physiologia Plantarum 70:381-388. https://doi.org/10.1111/j.1399-3054.1987.tb02832.x
Yin YG, Kobayashi Y, Sanuki A, Kondo S, Fukuda N, Ezura H … Matsukura C (2010). Salinity induces carbohydrate accumulation and sugar-regulated starch biosynthetic genes in tomato (Solanum lycopersicum L. cv. ‘Micro-Tom’) fruits in an ABA- and osmotic stress-independent manner. Journal of Experimental Botany 61:563-574. https://doi.org/10.1093/jxb/erp333
Zengin FK, Munzuroglu O (2005). Effects of some heavy metals on content of chlorophyll, proline and some antioxidant chemicals in bean (Phaseolus vulgaris L.) seedlings. Acta Biologica Cracoviensia. Series Botanica 47:157-164.
Zhishen J, Mengcheng T, Jianming W (1999). The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chemistry 64:555-559. https://doi.org/10.1016/S0308-8146(98)00102-2
Downloads
Published
How to Cite
Issue
Section
License
Papers published in Notulae Scientia Biologicae are Open-Access, distributed under the terms and conditions of the Creative Commons Attribution License.
© Articles by the authors; licensee SMTCT, Cluj-Napoca, Romania. The journal allows the author(s) to hold the copyright/to retain publishing rights without restriction.
License:
Open Access Journal - the journal offers free, immediate, and unrestricted access to peer-reviewed research and scholarly work, due SMTCT supports to increase the visibility, accessibility and reputation of the researchers, regardless of geography and their budgets. Users are allowed to read, download, copy, distribute, print, search, or link to the full texts of the articles, or use them for any other lawful purpose, without asking prior permission from the publisher or the author.













.png)















