Pollen grain expression of osmotic adjustment as a screening method on drought tolerance in several wine and table grape genotypes (Vitis vinifera L.)
DOI:
https://doi.org/10.15835/nsb12410843Keywords:
cytoplasm; grapevine clones; osmosis; plasmolysis; water deficitAbstract
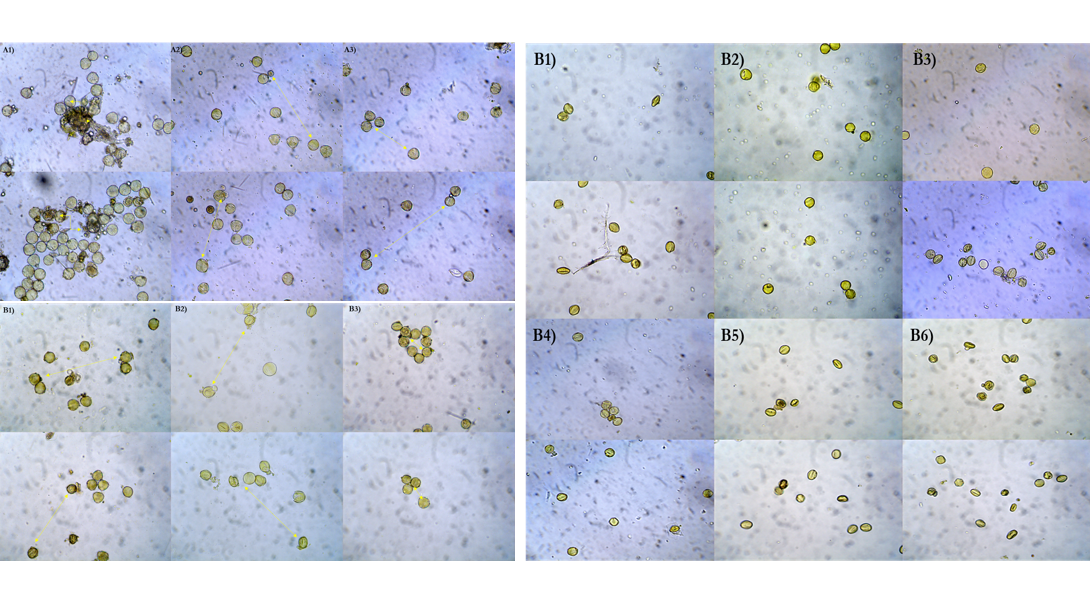
Osmotic adjustment is one of the important mechanisms to adapt to drought and it is the only one which is activated under any level of water stress in the plant cells. Grapevine pollen grains response was tested to osmotic stress in fourteen genotypes, initiated by immersion in 55% or 65% polyethylene glycol solutions without and with addition of potassium chloride, to estimate the expressions of osmotic adjustment. The pollen grain test found differences both in the measurements of projected area cytoplasm and expressions of osmotic adjustment present in the cells. Italian Riesling increased pollen grains cytoplasm in PEG solutions with added KCl much more than other genotypes and had the high values for both expressions of induced and overall osmotic adjustment. The results obtained for expression of induced osmotic adjustment underlined the high K+ accumulation capacity of ‘Italian Riesling’, ‘Burgund mare’ 86 Şt., ‘Muscat d’Adda’ 22 Şt., ‘Muscat Ottonel’ 16 Şt., ‘Pinot gris’ 14 Şt. and ‘Argessis’. The lack of correlation between expressions of induced and intrinsic osmotic adjustment indicated that induced osmotic adjustment expressed by K+ might use different mechanisms that are activated at the time of water stress with different levels of solute accumulation. Because the accumulation of K+ in the cells is important in all developmental stages and, in grape yield and quality, pollen responses to induced osmotic adjustment expressed by K+ could be used as a screening method, for establishing the level of drought sensitivity in the grape varieties under water stress.
Metrics
References
Bănică C, Petcu E, Giura A, Săulescu NN (2008). Relationship between genetic differences in the capacity of osmotic adjustment and other physiological measures of drought resistance in winter wheat (Triticum aestivum L.). Romanian Agricultural Research 25:7-11. https://www.researchgate.net/publication/268179519
Blum A (2009). Effective use of water (EUW) and not water-use-efficiency (WUE) is the target of crop yield improvement under drought stress. Field Crop Research 112:119-123. https://doi.org/10.1016/j.fcr.2009.03.009
Bora FD, Donici A, Oşlobeanu A, Fiţiu A, Babeş IC (2016). Qualitative assessment of the white wine varieties grown in Dealu Bujorului Vineyard, Romania. Notulae Botanicae Horti Agrobotanici 44(2):593-602. https://doi.org/10.15835/nbha44210434
Cardone MF, Perniola R, Catacchio CR, Alagna F, Rotunno S, Crupi P, … Bergamini C (2019). Grapevine adaptation to drought: New candidate genes for genotype-dependent response. BIO Web of Conferences 15, 01016, 42nd World Congress of Vine and Wine. https://doi.org/10.1051/bioconf/20191501016
Charrier G, Delzon S, Donnec JC, Zhang L, Delmas CEL, Merlin I, … Gambetta GA (2018). Drought will not leave your glass empty: Low risk of hydraulic failure revealed by long-term drought observations in worlds top wine regions. Science Advances 4. https://doi.org/10.1126/sciadv.aao6969
Chaves MM, Tenhunen JD, Harley P, Lange OL (1987). Gas exchange studies in two Portuguese grapevine cultivars. Physiologia Plantarum 70:639-647. https://doi.org/10.1111/j.1399-3054.1987.tb04318.x
Chaves MM, Santos TP, Souza CR (2007). Deficit irrigation in grapevine improves water-use-efficiency while controlling vigour and production quality. Annals of Applied Biology 150:237-252. https://doi.org/10.1111/j.1744-7348.2006.00123.x
Chaves MM, Zarrouk O, Francisco R, Costa JM, Santos T, Regalado AP, … Lopes CM (2010). Grapevine under deficit irrigation: hints from physiological and molecular data. Annals of Botany 105:661-676. https://doi.org/10.1093/aob/mcq030
Cuéllar T, Azeem F, Andrianteranagna M, Pascaud F, Verdeil JL, Sentenac H (2013). Potassium transport in developing fleshy fruits: the grapevine inward K+ channel VvK1.2 is activated by CIPK-CBL complexes and induced in ripening berry flesh cells. The Plant Journal 73:1006-1018. https://doi.org/10.1111/tpj.12092
David M (2009). Osmotic adjustment capacity and cuticular transpiration in several wheat cultivars cultivated in Algeria. Romanian Agricultural Research 26:29-33. https://www.researchgate.net/publication/266399528
David M (2012). Pollen grain expression of intrinsic and osmolyte induced osmotic adjustment in a set of wheat cultivars. Romanian Agricultural Research 29:45-52. https://www.researchgate.net/publication/266401970
Fan LM, Wang YF, Hong W, Wu WH (2001). In vitro Arabidopsis pollen germination and characterization of the inward potassium currents in Arabidopsis pollen grain protoplasts. Journal of Experimental Botany 52:1603-1614. https://doi.org/10.1093/jexbot/52.361.1603
Fuentes S, Sullivan W, Tilbrook J, Tyerman SD (2010). A novel analysis of grapevine berry tissue demonstrates a variety-dependent correlation between tissue vitality and shrivel. Australian journal of Grape Wine Research 13:66-71. https://doi.org/10.1111/j.1755-0238.2010.00095.x
Glăman G, Dejeu L, Brânduşe E, Şerdinescu A, Ion M (2018). Soiuri noi de viţă-de-vie şi portaltoi creaţi în România [New grapevine cultivars and rootstock genotypes obtained in Romania]. In: Glăman G, Dejeu L, Brânduşe E, Şerdinescu A, Ion M (Eds). Ampelografia României [Romanian Ampelography] IX. Ceres Bucureşti [Bucharest] pp. 63-64, 81-83, 185-186, 215-217, 395, 396, 397, 407, 408.
Hale CR (1977). Relation between potassium and the malate and tartare contents in grape berries. Vitis: Journal of Grapevine Research 16:9-19. https://doi.org/10.5073/vitis.1977.16.9-19
Hrazdina G, Parsons GF, Mattick LR (1984). Physiological and biochemical events during development and ripening of grape berries. American Journal of Enology and Viticulture 35:220-227. https://www.ajevonline.org/content/35/4/220
Kikuta SB, Richter H (1988). Rapid osmotic adjustment in detached wheat leaves. Annals of Botany 63(2):167-172. https://doi.org/10.1093/oxfordjournals.aob.a087647
Martins V, Cunha A, Gerós H, Hanana M, Blumwald E (2012). Mineral compounds in grape berry. In Gerós H, Chaves MM, Delrot S (Eds.). The biochemistry of the grape berry. Sharjah: Bentham Science pp. 23-43. https://doi.org/10.2174/97816080536051120101
Morgan JM (1980). Osmotic adjustment in the spikelets and leaves of wheat. Journal of Experimental Botany 31:655-665. https://doi.org/10.1093/jxb/31.2.655
Morgan JM (1983). Osmoregulation as a selection criterion for drought tolerance in wheat. Australian Journal of Agricultural Research 34:607-614. https://doi.org/10.1071/AR9830607
Morgan JM, Hare RA, Fletcher RJ (1986). Genetic variation in osmoregulation in bread and durum wheats and its relationship to grain yield in a range of field environments. Australian Journal of Agricultural Research 37:449-457. https://doi.org/10.1071/AR9860449
Morgan JM (1999). Pollen grain expression of a gene controlling differences in osmoregulation in wheat leaves: a simple breeding method. Australian Journal of Agricultural Research 50:953-962. https://doi.org/10.1071/AR98143
Mpelasoka BS, Schachtman DP, Treeby MT, Thomas MR (2008). A review of potassium nutrition in grapevines with special emphasis on berry accumulation. Australian Journal of Grape Wine Research 9:154-168. https://doi.org/10.1111/j.1755-0238.2003.tb00265.x
Moud AAM, Yamagishi T (2005). Application of project pollen area response to drought stress to determine osmoregulation capability of different wheat (Triticum aestivum L.) cultivars. International Journal of Agriculture and Biology 7(4):604-605.
Mouline K, Véry AA, Gaymard F, Boucheren J, Pilot G, Devic M, … Sentenac H (2002). Pollen tube development and competitive ability are impaired by disruption of a Shaker K+ channel in Arabidopsis. Genes&Development 16:339-350. https://doi.org/10.1101/gad.213902
Oprea A, Indreaş A (2000). Floarea şi polenul [The flower and pollen]. In: Oprea A, Indreaş A (Eds). Viticultura [Viticulture]. Ceres Bucureşti [Bucharest] pp.63-64.
Parker WC, Pallardy SG (1985). Genotypic variation in tissue water relations of leaves and roots of black walnut (Juglans nigra) seedlings. Physiologia Plantarum 64:105-110. https://doi.org/10.1111/j.1399-3054.1985.tb01219.x
Patil BS, Ravikumar RL (2011). Osmotic adjustment in pollen grains: a measure of drought adaptation in sorghum? Current Science 100(3):377-382. https://www.researchgate.net/publication/257299541
Rehman S, Rha ES, Ashraf M, Lee KJ, Yun SJ, Kwak YG (2004). Does barley (Hordeum vulgare L.) pollen swell in fractions of a second? Plant Science 167:137-142. https://doi.org/10.1016/j.plantsci.2004.03.013
Rodrigues ML, Chaves MM, Wendler R, David MM, Quick WP, Leegood RC, … Pereira JS (1993). Osmotic adjustment in water stressed grapevine leaves in relation to carbon assimilation. Australian Journal of Plant Physiology 20:309-321. https://doi.org/10.1071/PP9930309
Rogiers YS, Greer DH, Hatfield JM, Orchard BA, Keller M (2006a). Mineral sinks within ripening grape berries Vitis vinifera (L.). Vitis-Geilweilerhof 45(3):115-123. https://www.researchgate.net/publication/282500219
Rogiers YS, Zelmari AC, Walker RR, Deloire A, Tyerman SD (2017). Potassium in the grape (Vitis vinifera L.) berry: transport and function. Plant Science 8:1629. https://doi.org/10.3389/fpls.2017.01629
Rossdeutsch L, Edwards E, Cookson SJ, Barrieu F, Gambetta GA, Delrot S, Ollat N (2016). ABA-mediated responses to water deficit separate grapevine genotypes by their genetic background. BMC Plant Biology 16:91. https://doi.org/10.1186/s12870-016-0778-4
Schultz HR (2003). Differences in hydraulic architecture account for near-isohydric and anisohydric behavior in two field-grown Vitis vinifera L. cultivars during drought. Plant Cell and Environment 26:1393-1405. https://doi.org/10.1046/j.1365-3040.2003.01064.x
Serpe M, Matthews M (2000). Turgor and cell wall yielding in dicot leaf growth in response to changes in relative humidity. Australian Journal of Plant Physiology 27:142-147. https://doi.org/10.1071/PP00055
Serra I, Strever A, Myburgh PA, Deloire A (2014). Review: the interaction between rootstocks and cultivars (Vitis vinifera L.) to enhance drought tolerance in grapevine. Australian Journal of Grape and Wine Research 20:1-14. https://doi.org/10.1111/ajgw.12054
Soar CJ, Speirs J, Maffei SM, Penrose AB, McCarthy MG, Loveys BR (2006). Grape vine varieties Shiraz and Grenache differ in their stomatal response to VPD: apparent links with ABA physiology and gene expression in leaf tissue. Australian Journal of Grape and Wine Research 12:2-12. https://doi.org/10.1111/j.1755-0238.2006.tb00038.x
Sobrado MA (1986). Aspects of tissue water relations and seasonal changes of leaf water potential components of evergreen and deciduous species coexisting in tropical dry forest. Oecologia 68:413-416. https://doi.org/10.1007/BF01036748
Sobrado MA (1990). Drought response of tropical corn. 2. Leaf expansion rate and water relations under controlled conditions. Maydica 35:227-234. https://www.cabdirect.org/cabdirect/abstract/19901617342
Steel RG, Torrie JH, Dickey DA (1997). Principles and procedures of statistics: a biometrical approach, 3rd. WCB MCgraw-Hill, Boston.
Tillbrook J, Tyerman SD (2008). Cell death in grape berries varietal differences linked to xylem pressure and berry weight loss. Functional Plant Biology 35:173-184. https://doi.org/10.1071/FP07278
Tschaplinski TJ, Blake TJ (1989). Water-stress tolerance and late-season organic solute accumulation in hybrid poplar. Canadian Journal of Botany 67(6):1681-1688. https://doi.org/10.1139/b89-212
Turner NC, Jones MM (1980). Turgor maintenance by osmotic adjustment: a review and evaluation. In: Turner NC, Kramer PJ (Eds). Adaptation of plants to water and high temperature stress. John Wiley & Sons London pp 87-103.
Zhao LN, Shen LK, Zhang WZ, Zhang W, Wang Y, Wu WH (2013). Ca2+-dependent protein kinase 11 and 24 modulate the activity of the inward rectifying K+ Channels in Arabidopsis pollen tubes. The Plant Cell 25:649-661. https://doi.org/10.1105/tpc.112.103184
Downloads
Published
How to Cite
Issue
Section
License
Papers published in Notulae Scientia Biologicae are Open-Access, distributed under the terms and conditions of the Creative Commons Attribution License.
© Articles by the authors; licensee SMTCT, Cluj-Napoca, Romania. The journal allows the author(s) to hold the copyright/to retain publishing rights without restriction.
License:
Open Access Journal - the journal offers free, immediate, and unrestricted access to peer-reviewed research and scholarly work, due SMTCT supports to increase the visibility, accessibility and reputation of the researchers, regardless of geography and their budgets. Users are allowed to read, download, copy, distribute, print, search, or link to the full texts of the articles, or use them for any other lawful purpose, without asking prior permission from the publisher or the author.













.png)















