The risk mitigation indices associated with some risk mitigation protocols performed on contaminated aqua-cultured catfish (Clarias gariepinus)
DOI:
https://doi.org/10.15835/nsb12110633Keywords:
bio-risk; cross-contamination; multidrug-resistant AmpC beta-lactamase-producing Enterobactercloacae pathogenAbstract
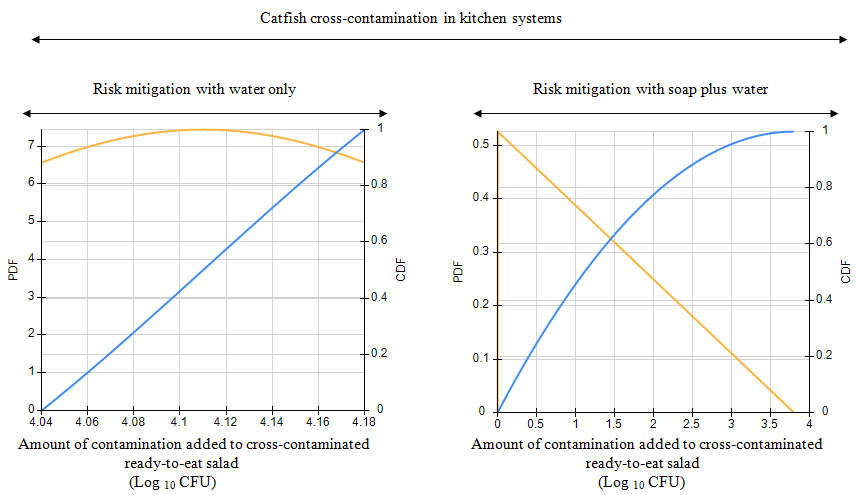
The present research quantified the risk mitigation indices associated with some risk mitigation protocols practiced by humans in Nigeria to mitigate the baseline risk of illness that were associated with Clarias gariepinus contaminated with multidrug-resistant AmpC beta-lactamase-producing Enterobacter cloacae pathogen. Identification of the multidrug-resistant AmpC beta-lactamase-producing E. cloacae pathogen was performed with phenotypic and molecular methods. The United States FDA-iRisk stochastic software quantified the baseline and residual risk of illness associated with the contamination and cross-contamination routes of the C. gariepinus chain. The fresh adult C. gariepinus was contaminated with the multidrug-resistant E. cloacae pathogen (median concentration = 4.49 CFU g-1), but this was significantly reduced by the risk mitigation protocols practiced by the human consumers. The risk mitigation practices of human consumers resulted in a risk mitigation index of 0.92, which was equivalent to a 92% reduction in the baseline risk of illness (baseline risk of illness = 7.58 × 10-4; residual risk of illness = 5.9 × 10-5). In spite of the significant risk mitigation, inducible AmpC beta-lactamase resistance genes was still found in the residual multidrug-resistant Enterobacter cloacae pathogen, which survived the inadequate risk mitigation protocols that accounted for 8% residual risk of illness.
Metrics
References
Adedeji OB, Onwenefah M (2013). The antibiotic resistant patterns of bacterial flora of cultured catfish fed with poultry hatchery waste from selected farms in Ibadan, Nigeria. Researcher 5:37-43.
Aven T (2016). Risk assessment and risk management: Review of recent advances on their foundation. European Journal of Operational Research 253:1-13.
Barker GC (2002). Risk assessment for Clostridium botulinum: a network approach. International Biodeterioration and Biodegradation 50:167-175.
Boss R, Overesch G, Baumgartner A (2016). Antimicrobial resistance of Escherichia coli, Enterococci, Pseudomonas aeruginosa, and Staphylococcus aureus from raw fish and seafood imported into Switzerland. Journal of Food Protection 79:1240-1246.
Cassin MH, Paoli GM, Lammerding AM (1998). Simulation modelling for microbial risk assessment. Journal of Food Protection 61:1560-1566.
Clinical and Laboratory Standards Institute (CLSI) (2014). Performance standards for antimicrobial susceptibility testing. Twenty-fourth informational supplement, CLSI document M100-S24, Wayne, PA, United States pp 50-57.
Codex Alimentarius Commission (1999). Principles and guidelines for the conduct of microbiological risk assessment. CAC/GL-30, Food and Agricultural Organization of the United Nations, Rome. http://www.fao.org/3/y1579e/y1579e05.htm.
Conceicao T, Faria N, Lito L, Cristino JM, Salgado JM, Duarte A (2004). Diversity of chromosomal AmpC L-lactamases from Enterobacter cloacae isolates in a Portuguese hospital. FEMS Microbiology Letters 230:197-202.
Efuntoye MO, Olurin KB, Jegede GC (2012). Bacterial flora from healthy Clarias gariepinus and their antimicrobial resistance pattern. Advanced Journal of Food Science and Technology 4:121-125.
Ekundayo FO, Diyaolu DO, Fasakin EA (2014). Composition, distribution and antibiotic sensitivities of bacteria associated with cultured Clarias gariepinus (Burchell 1822). Malaysia Journal of Microbiology 10:72-79.
FDA, EPA (2017). Eating fish: what pregnant women and parents should know. www.fda.gov/fishadvice and www.epa.gov/fishadvice .
Food and Drug Administration, Center for Food Safety and Applied Nutrition, Joint Institute for Food Safety and Applied Nutrition, Risk Sciences International (FDA/CFSAN/JIFSAN/RSI) (2017). FDA-iRISK® version 4.0 Technical Document. College Park, Maryland, United States.
Imade OS, Akinnibosun FI (2018). The Nigerian catfish (Clarias gariepinus) pisciculture systems: reservoirs of multidrug-resistant bacteria. Journal of Applied Microbiology 125:1552-1568.
Jackson TC, Hardin MD, Acuff GR (1996). Heat resistance of E. coli O157:H7 in a nutrient medium and in ground beef E. coli O157:H7 in a nutrient medium and in ground beef. Food Protection 59:230-237.
Karch H, Meyer T (1989). Single primer pair for amplifying segments of distinct shiga-like-toxin genes by polymerase chain reaction. Journal of Clinical Microbiology 27:2751-2757.
Kirk MD, Angulo FJ, Havelaar AH, Black RE (2017). Diarrhoeal disease in children due to contaminated food. Bulletin of the World Health Organization 95:233-234.
Lane DJ (1991). 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (Eds), Nucleic acid techniques in bacterial systematics. John Wiley and Sons, New York, USA pp 115-175.
Park MS, Cho JI, Lee SH, Bahk GJ (2014). A study on dose-response models for foodborne disease pathogens. Journal of Food Hygiene and Safety 29:299-304.
Pérez-Pérez FJ, Hanson DH (2002). Detection of plasmid-mediated AmpC β-lactamase genes in clinical isolates by using multiplex PCR. Journal of Clinical Microbiology 40:2153-2162.
Pires SM, Fischer-Walker CL, Lanata CF, Devleesschauwer B, Hall AJ, Kirk MD (2015). Aetiology-specific estimates of the global and regional incidence and mortality of diarrhoeal diseases commonly transmitted through food. PLoS ONE 10:e0142927.
Public Health England (2014). Preparation of samples and dilutions, plating and sub-culture. In: Microbiology services food water and environmental microbiology standard method. FNES26 (F2), London pp 12-13.
Qadri F, Saha A, Ahmed T, Al Tarique A, Begum YA, Svennerholm AM (2007). Disease burden due to enterotoxigenic Escherichia coli in the first 2 years of life in an urban community in Bangladesh. Infection and Immunity 75:3961-3968.
Rosenberger WF, Lachin JM (2002). Randomization in Clinical Trials Theory and Practice. John Wiley and Sons pp 288.
Surendran SPK, Thampuran N (2009). PCR-based detection of enterotoxigenic isolates of B. cereus from tropical seafood. Indian Journal of Medical Research 129:316-320.
Downloads
Published
How to Cite
Issue
Section
License
Papers published in Notulae Scientia Biologicae are Open-Access, distributed under the terms and conditions of the Creative Commons Attribution License.
© Articles by the authors; licensee SMTCT, Cluj-Napoca, Romania. The journal allows the author(s) to hold the copyright/to retain publishing rights without restriction.
License:
Open Access Journal - the journal offers free, immediate, and unrestricted access to peer-reviewed research and scholarly work, due SMTCT supports to increase the visibility, accessibility and reputation of the researchers, regardless of geography and their budgets. Users are allowed to read, download, copy, distribute, print, search, or link to the full texts of the articles, or use them for any other lawful purpose, without asking prior permission from the publisher or the author.













.png)















